Search OneLab Network Past Events
Risk Assessment in Clinical Laboratories
When Ebola entered the United States in 2014, healthcare workers were faced with the very real threat of a deadly disease presenting itself anywhere at any time. This revealed the importance of needing to be prepared and ready to respond.

Managing Burnout in a Post-COVID World
Managing burnout in a post-COVID world is essential as burnout can lead to reduced productivity, increased absenteeism, and higher turnover rates.
Empowering Healthcare: Bridging Gaps, Building Futures with the Clinical Laboratory Development Program
The shortage of clinical diagnostic laboratory personnel directly impacts the quality of healthcare services, patient outcomes, and the healthcare system's ability to respond to emerging health challenges and technological advancements effectively.
How to Plan for B. pseudomallei Exposure Cases - A Laboratory Perspective
Melioidosis is an infectious disease caused by the bacteria Burkholderia pseudomallei.

CLIA Proficiency Testing (PT) Final Rule, CMS-3355-F
This presentation will provide information on the CLIA Proficiency Testing (PT) Final Rule which was published in the Federal Register Notice on July 11, 2022.
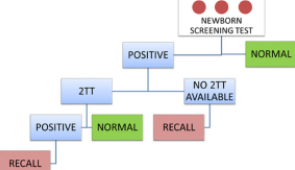
Oregon State Public Health Laboratory Shares Lessons Learned from Its Newborn Screening Program
The history of newborn screening (NBS) demonstrates the strength and integrity of its mission: to identify children with congenital conditions who need treatment to prevent adverse outcomes.

Biosafety Practices and Reporting Occupational Exposures for Select Agents and Toxins (Part 2)
This basic-level webinar is designed to help clinical and veterinary laboratory professionals understand how to report select agent and toxin identifications and recognize and report exposure incidents.

Recognizing, Identifying, and Reporting the Identification of Select Agents and Toxins
This beginner-level webinar is for clinical and veterinary laboratory professionals to understand how to report select agent and toxin identifications and recognize and report exposure incidents.
The Survey Process: What You Need to Know for Your CLIA Survey
Preparing for laboratory survey? Laboratory staff are invited to join this webinar, where Marranda Scott, a Clinical Laboratory Scientist with the CDC Division of Laboratory Systems, will review processes and requirements for a CLIA laboratory survey.
